Heating Treating Solutions

Industrial Gases from a Global Leader
- Global leading manufacturer of nitrogen, oxygen, and argon
- World leading producer and supplier of hydrogen and helium
- Reliable supply of industrial gases
- Excellent record of meeting on-time needs
- Industry leader in safety
- Technical support when you need it
- World-class customer service
Ask the Expert

Liang He
Advanced Materials Processing R&D Engineer, North America
Bright annealing of steels requires conditions that are reducing to steel oxides. Traditionally, the Ellingham diagram has been used to predict the conditions that correspond to oxidation of pure metals or reduction of their oxides. This method can be used to predict the conditions that should be reducing to iron oxides and the oxides of the alloying elements added to steels, such as chromium when stainless steels are considered. This traditional approach is not precise because it only uses thermodynamic data for pure metals and their oxides—it ignores the fact that iron and alloying elements form a solid solution. In addition, you can only determine the approximate equilibrium partial pressure ratio of hydrogen and water vapor for oxidation of a specific metal at a particular temperature.
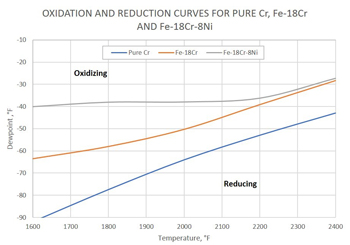
Alternatively, you can use more accurate and convenient diagrams for steels and other alloys, which are created with the help of modern databases and computer programs, such as FactSage™ (thermochemical software and database package developed jointly between Thermfact/CRCT and GTT-Technologies) or Thermo-Calc software. Using the oxidation-reduction curves, presented as dew point of pure hydrogen or nitrogen-hydrogen atmospheres versus temperature, you can quickly select the atmosphere for annealing steels without formation of oxides. The diagram in Figure 1 was calculated using FactSage. This diagram shows that oxidation-reduction curves for Fe-18%Cr and Fe-18%Cr-8%Ni systems representing stainless steels are higher than the corresponding Cr/Cr₂O₃ curves. For alloys (e.g. steels), you can achieve more precise calculations using thermodynamic data from both the pure substances (i.e. pure metals and oxides) and solutions databases. Such diagrams can be produced specifically for the steels of interest and variety of atmosphere compositions.
These methods can help you troubleshoot and optimize your annealing operation by balancing hydrogen usage versus product quality.
Oxidation-reduction curves for pure chromium, Fe-18%Cr and Fe-18%Cr-8%Ni for the total pressure of 1 atm with a hydrogen partial pressure of 0.05 atm, corresponding N₂-5% H₂. (This diagram has been produced using thermodynamic data for solutions in addition to the data for pure elements and their oxides.)
Figure 1:
CASE STUDY

Each Atmosphere Solution is Customized for Your Needs
We can help you choose the right atmosphere composition.
- Select and provide blending and flow control equipment
- Perform on-site atmosphere analyses for consistent dimensional control, carbon control, and physical and metallurgical properties
- Optimize your furnace process to lower operating costs
- Perform state-of-the-art metallographic and chemical analyses (SEM, ESCA, SAM, ISS, SIMS)
- Provide safety training
- Test atmospheres using your parts in our lab furnaces
- Demonstrate our system at your facility

Industrial Gas Audits and Leak Detection Services
Gases
Argon
Compressed argon gas and liquid argon in a variety of purities and in various modes of supply around the world thanks to our network of storage and transfill facilities.
Helium
An inert gas for cryogenic, heat transfer, shielding, leak detection, analytical and lifting applications
Hydrogen
Valued for its reactive and protective properties, and used by many industries such as electronics, foods, glass, chemicals, refining and more can benefit from its unique properties to improve quality, optimize performance and reduce costs.
Nitrogen
Useful as a gas, for its inert properties, and as a liquid for cooling and freezing. Virtually any industry can benefit from its unique properties to improve yields, optimize performance and make operations safer.
Get in touch with our technical experts.
Contact us to put decades of annealing experience to work for you.
